Electronics goods maker Sharp Corporation has developed a device equipped with Plasmacluster technology that it claims has shown, in a research test, ability to reduce the infectious titer of the novel coronavirus (COVID-19) by more than 90 percent.
The product could be used in hospitals, malls, theatres, offices as well as homes. India has reported a total of 62,25,764 COVID-19 positive cases. A total of 97,497 people have lost their lives to COVID-19 in India so far.
Infectious titer is the concentration of viral particles that can transduce cells. Here, transduction is the process by which foreign DNA is introduced into a cell by a virus or viral vector.
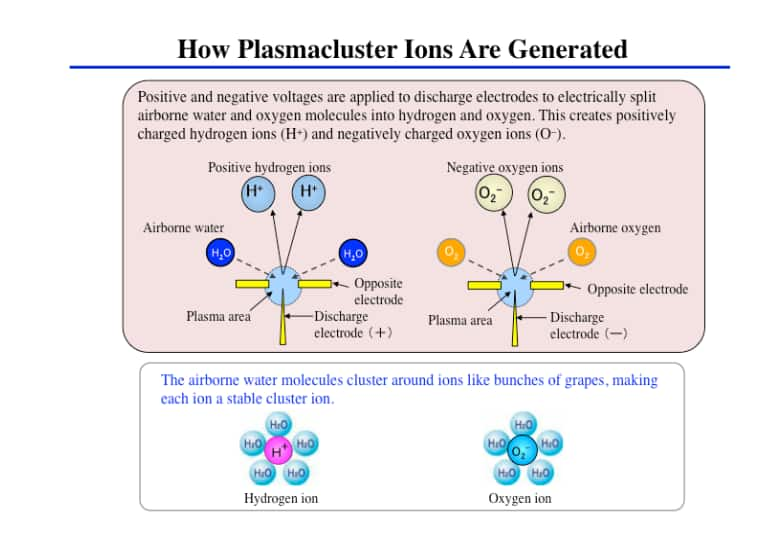
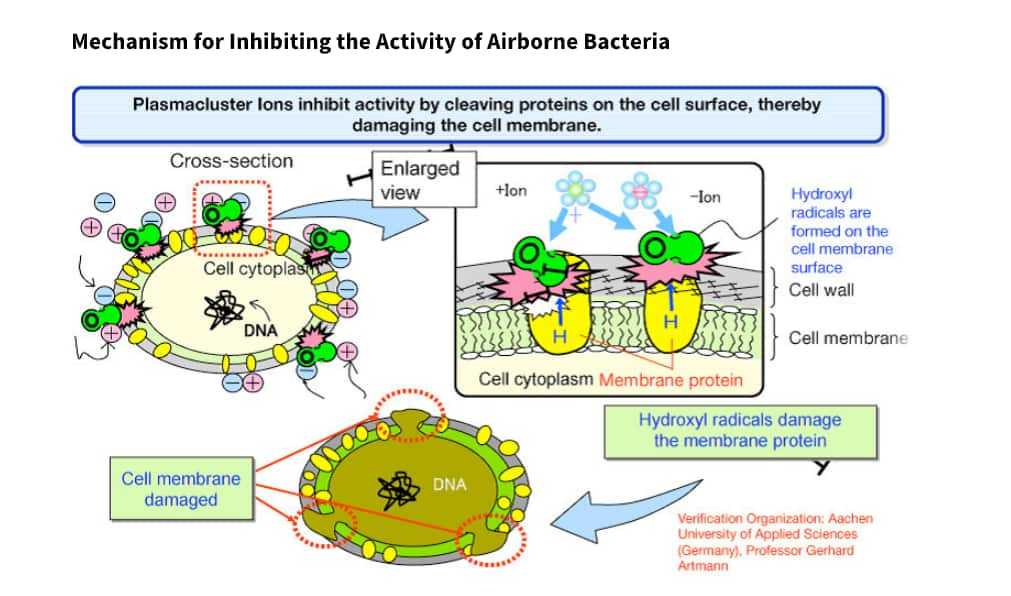
Plasmacluster technology is where positively charged hydrogen and negatively charged oxygen ions are released into the air simultaneously, and the positive and negative ions instantaneously bond on the surface of airborne bacteria, fungi, viruses, allergen. This is an air purification technology that works to suppress the activity of bacteria, by breaking down proteins on their surface by a chemical reaction.
This technology inhibits activity by breaking the protein on the cell surface which damages the cell membrane. This could effectively kill the virus.
In this study, the virus testing device was equipped with Plasmacluster technology. Then the virus was passed through the test device from a generator.
Later, the aerosolized virus was recovered after exposure to Plasmacluster ions. Then the test calculated the infectious virus titer of the recovered virus solution by a plaque assay. Assay means a standard testing to evaluate the number of infectious virus in the sample.
The test, the company said, demonstrated that the virus infectious titer (number of the virus) was reduced more than 90 percent.
Sharp Corporation said in a statement that this test was accomplished in collaboration with Professor Jiro Yasuda of the National Research Center for the Control and Prevention of Infectious Diseases/Institute of Tropical Medicine, Nagasaki University, and Professor Asuka Nanbo (a board member of the Japanese Society for Virology) of the same institution.
The other collaborators were Professor Hironori Yoshiyama of the Department of Microbiology, Shimane University, Faculty of Medicine (also, a board member of the Japanese Society for Virology), and Nagasaki University, an internationally respected authority on infectious diseases research.
The outbreak of coronavirus disease (COVID-19) caused by the novel coronavirus (SARS-CoV-2) was first reported in December 2019.
Sharp said that in 2004, the company used Plasmacluster technology against feline (cat) coronavirus, a member of the Corona viridae family. In the following year of 2005, Sharp also demonstrated its effectiveness against the original SARS coronavirus (SARS- CoV), which caused the outbreak of 2002-2003 and is also genetically similar to the novel coronavirus (SARS-CoV-2).
Dr Jiro Yasuda, Professor of National Research Center for the Control and Prevention of Infectious Diseases, Nagasaki University, said, "Disinfectants such as alcohol and detergents (surfactants) are well-known to be effective to reduce the risk of the virus on materials. However, for infection via aerosols (micro-droplets), there are few effective countermeasures such as a mask. Today, we demonstrated the effective inactivation of SARS-CoV-2 in airborne droplets by using Plasmacluster technology. We expect it would be useful to reduce the risk of infection in real spaces like offices, homes, medical facilities and vehicles too.”
While the device is not yet commercially available, Sharp is in talks to partner with Indian firms to offer this product in India. This could be used in commercial establishments and homes.
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!
Find the best of Al News in one place, specially curated for you every weekend.
Stay on top of the latest tech trends and biggest startup news.