


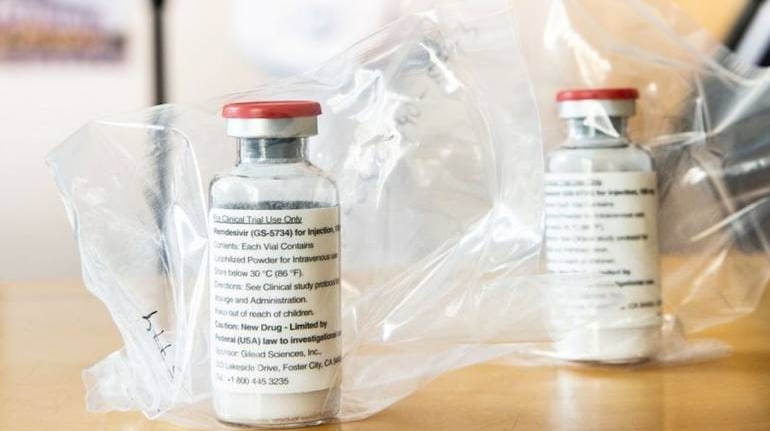
Drug maker Cipla said on Tuesday it is scaling up the production of antiviral drug remdesivir, used in COVID-19 treatment, by manufacturing the drug at its Goa plant.
"Currently the demand is huge for remdesivir, and we are scaling up our internal manufacturing of Remdesivir," Kedar Upadhye, global chief financial officer of Cipla, told Moneycontrol.
Upadhye did not specify the new capacity that will be added, but hinted that over a period of time, the company wants to move remdesivir production in-house.
Cipla has partnered with BDR Pharmaceuticals for API supplies and Sovereign Pharma for finished dosages.
Sovereign Pharma has the capacity to produce up to 95,000 vials per month.
Cipla launched remdesivir under brand name Cipremi at Rs 4,000 per vial in July. Cipremi is the cheapest of all generic versions of remdesivir available in the market.
A patient needs five vials of the drug.
Cipla is among the six Indian generic pharma manufacturers that have signed non-exclusive voluntary licencing agreements with Gilead Sciences for its patented drug Veklury (remdesivir).
Moneycontrol reported that 800,000 doses of remdesivir are expected to be available in the country in August as existing suppliers expand capacity, and new manufacturers get ready to enter the market with their generic versions.
Tocilizumab shortages
Besides remdesivir, another drug tocilizumab that Cipla markets under licensing pact with Swiss pharma company Roche is also seeing huge demand, leading to shortage of the drug in the market.
"For tocilizumab, unfortunately we are not able to do anything. But we are in touch with them (Roche) to ease supplies," said Upadhye.
Upadhye further said nobody was prepared for this kind of bump up in demand, and it is difficult for them to increase the capacity.
Upadhye said the company has the broadest range of drugs and products for COVID-19, from face masks to sanitizers and drugs like remdesivir, favipiravir, tocilizumab, hydroxychloroquine, azithromycin, vitamins, among others.
"The idea is to obviously service patients. Some of these are in-license products. The margins are low. Whatever little margin we make is actually invested in giving PPE kits and other things to doctors.
"The retained profits of COVID-19 products are hardly anything. In fact, it will be negative also, if I start making a list," Upadhye said.
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!
Find the best of Al News in one place, specially curated for you every weekend.
Stay on top of the latest tech trends and biggest startup news.