


The Co-WIN portal – www.cowin.gov.in – faced minor glitches on March 1, just as registration for the second phase of the nationwide vaccination drive commenced. Starting at 9.00 am, eligible beneficiaries were able to register on the Co-WIN 2.0 portal for immunisation.
Everyone above the age of 60 years and those over 45 years with comorbidities will be able to get the COVID-19 vaccine for free at government facilities and for a charge at a number of private hospitals. Private hospitals have been allowed to charge up to Rs 250 per dose of the COVID-19 vaccine.
Co-WIN, a digital platform, was created for real-time monitoring of COVID-19 vaccine delivery. The new version of the Co-WIN platform is GPS-enabled and beneficiaries will have the option to choose the inoculation session site both at the government and private facilities.
However, some people noted that they did not receive an OTP (one-time-password) upon entering the mobile number as part of the registration process. Some people said they received the OTP after a delay.
Some users were shown the message: "CoWIN server is facing issues. Please try later."
The Union Health Ministry also clarified that registration and booking of appointments will be done only through the Co-WIN portal and that there is no app for beneficiaries. The app on Play Store is only for administrators, the ministry said.
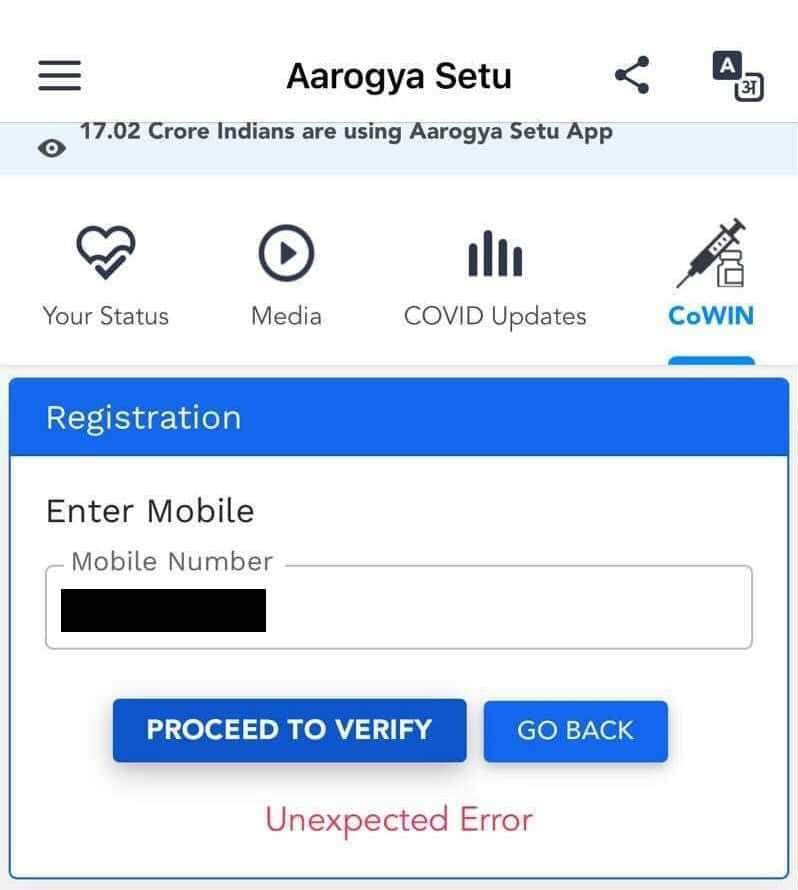 Screenshot of the Aarogya Setu app showing an "unexpected error" while registering in the Co-WIN section
Screenshot of the Aarogya Setu app showing an "unexpected error" while registering in the Co-WIN section
Follow our LIVE blog for the latest updates of the coronavirus pandemic
More than 1.42 crore beneficiaries have received at least their first jabs as part of the two-dose regime.
As of February 28, India had recorded more than 1.1 crore confirmed COVID-19 cases, including 1.57 lakh deaths. A total of 1.07 crore patients had recuperated from the infectious disease. However, more than 1.64 lakh cases remained active across the country, comprising 1.48 percent of the total caseload. India's recovery rate stood at 96.1 percent even as many states were witnessing a resurgence in the number of cases being reported per day.
COVID-19 Vaccine Tracker: All you need to know about manufacturing and pricing
A speedy and efficient vaccination drive is being seen as the only main way to stop the spread of the disease and to restore normalcy in the pandemic-battered global economy.
In early January, India’s drug regulator had approved two vaccines - Covaxin developed by Bharat Biotech and Covishield from the Oxford/AstraZeneca stable being manufactured by the Serum Institute of India (SII) - for emergency use in the country. It is expected that more vaccine candidates will get emergency use approval in the coming months.
Click here for Moneycontrol’s full coverage of the coronavirus pandemic
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!