


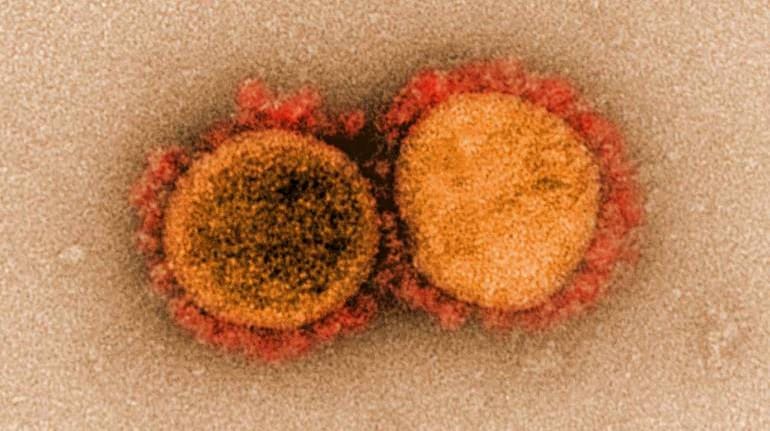
Of all the pandemic questions bedeviling scientists, the one that Juan Perilla is asking might be among the strangest: If a shrunk-down hand were to squeeze the coronavirus, would it squish, or would it shatter?
Viruses like HIV tend to be on the softer side, smooshing down like a foam ball, whereas the ones that cause influenza are more brittle, prone to cracking like an egg, said Perilla, a biophysical chemist at the University of Delaware in Newark. Coronaviruses, he suspects, are somewhere in the middle, a sort of tactile Goldilocks in the world of infectious disease.
“It’s something you never consider when you talk about viruses,” Perilla said. But it’s part and parcel, he added, of “trying to understand how a virion is strung together.”
Like many other microbes, viruses are known best as malady-toting motes of misfortune — obvious grist for biologists keen to understand the inner workings of infection. But in recent years, physicists too have joined the field, eager to decipher how viruses cobble themselves together and move from place to place despite lacking most of the machinery that enables cells to replicate and run.
Some physicists, like Perilla, are teasing apart the mechanical properties of virus particles, while others monitor the forces that bring the bits and pieces of viruses together or tear them apart. Some are even enlisting physics to design the next generation of COVID-19 treatments, whether by disrupting the forces that assemble viruses inside human cells or by creating a suite of self-replicating vaccines.
“Once you understand how something works, you can understand how to sabotage it,” said Jodi Hadden-Perilla, a biophysical chemist at the University of Delaware. (Perilla and Hadden-Perilla, a married couple, are professional collaborators.)
Physics and virology are now so academically intertwined that their union has acquired a formal name: virus physics (or, to some, physical virology). And the data its pioneers produce can have consequences far beyond the microbial world.
“At the end of the day,” said LaNell Williams, a virus expert and physicist at Harvard University, “we’re trying to figure out what physics viruses already know.”
Simply complex
Viruses are both absurdly simple and dizzyingly complex. Many are composed of little more than a tangle of genetic material stuffed into a protein coat — not even enough for some scientists to consider them alive. They are entirely dependent on the inner workings of cells, and cannot make more of themselves without help from the life-forms they infect.
And yet, under the right conditions, basic viral components can accomplish what many natural phenomena cannot: a process called self-assembly, combining their bits into neat, structured particles without any external forces to guide them, like cake ingredients mixing themselves into batter or snowflakes sprouting spontaneously out of room-temperature water.
It’s a beguiling question, Williams said: “Why do ordered things form without any assistance?”
Scientists can recreate this mysterious process in laboratory test tubes. They swirl together hunks of genetic material and proteins in salty chemical soup, and watch the viruses erupt forth. “It’s remarkable,” said Vinothan Manoharan, a biophysicist and engineer at Harvard University and Williams’ adviser. “You have these pieces spontaneously coming together, without any active intervention: It just happens.”
Bill Gelbart, a physical-chemist-turned-virologist at the University of California, Los Angeles, said it was this astounding buildability that drew him to viruses 20 years ago — an unexpected midcareer transition. Other diseases, like cancer, could not be boiled down to their base components and assembled or disassembled at will. Even bacteria, some of the simplest and best understood life-forms, can’t reform once ripped apart.
Viruses are different. “I was powerfully fascinated by the idea that an infectious virus particle can be made from scratch from purified components,” Gelbart said. “I think the Dr. Frankenstein in me got the better of me. I thought, ‘I have to do that.’”
That same desire has seeded dozens of virus physics labs around the globe (several of which cite Gelbart as an inspiration). Manoharan’s lab is one, focused on the assembly of bacteriophages, viruses that exclusively infect bacteria.
The static pictures and diagrams in textbooks don’t do viruses justice, Manoharan said. They tend to focus on the products of virus assembly. But the pieces within viruses — the genomes and proteins — start out as a dynamic jumble, and the chaotic motions are an obstacle for any virus that might organize itself into existence. Moreover, its components must find each other, without mistaking the cell’s cargo for its own.
By and large, researchers don’t yet have answers for how viruses solve this packaging problem. The process is especially complex for coronaviruses, which possess some of the largest known RNA-based genomes known to science. (RNA is a single-stranded cousin of the DNA that encodes the human genome.)
But scientists have already figured out ways to spy on the coronavirus’ assembly process, and start to replicate it for themselves.
A tight squeeze
A crucial early step in the coronavirus’ construction is carried out by a protein called nucleocapsid, which wrangles the virus’ RNA into a tight conformation and holds it in place, so it can be more easily packaged.
Jasmine Cubuk, a biochemist and biophysicist at Washington University in St. Louis, is using a microscopy technique called fluorescence resonance energy transfer, or FRET, to monitor these molecular tangos in real time. In a study that has not yet been published in a scientific journal, Cubuk and her colleagues showed that nucleocapsids are wiggly, which might help them shimmy around a host cell in search of their viral RNA partners.
Cubuk compared the flexibility of nucleocapsids to fly casting, where the compliance of the line that’s cast yields “a larger capture radius” and makes it easier to snare the target.
Once nucleocapsid and RNA have partnered up, they cloister themselves from the surrounding molecules, like globs of oil separating out of a salad vinaigrette, Cubuk and her colleagues found. These movements appear to create concentrated pockets of viral material, and may help explain how the vast genome of the coronavirus “gets packaged into something so tiny,” Cubuk said.
At the University of California, Riverside, a team led by physicist Roya Zandi has turned its lens to the next step of the process: bundling the virus’ newly condensed genome into its fatty, fragile outer coat, called the envelope. To accomplish this, the virus must steal some of its host cell’s greasy membranes, while interlacing proteins of its own. Zandi and her colleagues, using computational models and simulations, are testing how human and virus ingredients come together.
One point of fascination, she said, is how the virus forces its outer packaging to curve around it, transforming the pathogen into an intricate, spike-studded sphere. “The membrane has to bend quite a lot around such a big genome,” she said. “What kind of interactions between proteins can induce that?”
The knowledge contained within these experiments can teach scientists not just how to build viruses, but how to destroy them as well.
Breaking a virus
Decades of work have reaffirmed that the virus assembly process is extraordinarily fickle. Tweak one variable and the whole contraption falls apart, or never forms.
That frailty is exactly what many researchers are counting on. Adam Zlotnick, a biophysicist at Indiana University Bloomington, specializes in disrupting virus assembly. An obvious strategy, he said, would be to introduce a drug or other treatment to slow or stop the building of new viruses. But he and his colleagues have found that using drugs to speed up the process can also prove disastrous. Unable to wriggle into the proper configuration, or to correct initial errors, the virus’ bits and pieces might glue together improperly, layering mistakes atop mistakes until the entire structure is malformed.
“If you make it go a little bit faster, that’s bad, you get more virus,” said Zlotnick, who compared the acceleration to the chaos that might unfold if an assembly line was moving too fast. “But a lot faster? It’s going to screw up and make defective particles.”
Zlotnick has spent much of his career working on the hepatitis B virus, and has helped to develop drugs that can foil the pathogen’s assembly process, some of which are now in clinical trials. Although the coronavirus is a very different beast, it could someday be vulnerable to the same general strategy, Zlotnick said.
Other researchers are eyeing another step in the virus-building pipeline: the generation of virus genes, before they are packaged into their protein capsules. Carlos Bustamante, a biophysicist at the University of California, Berkeley, is set on sabotaging a protein called polymerase, which copies the coronavirus’ genome.
The polymerase zooms over a stretch of RNA and replicates it letter for letter, a process that requires an intimate connection between the molecules and enough force to propel the protein. That force can be measured with a tiny set of “optical tweezers” — a laser that hooks on to one end of the polymerase, using a microscopic glass bead, and pulls in the direction opposite the protein’s path. “We are playing tug of war,” Bustamante said. “Every time it moves, it has to pull us.”
The hope, he said, is to understand the tug well enough to design a drug that blocks the RNA-copying process.
In Delaware, Perilla and Hadden-Perilla are studying a moment even earlier in the process, when the coronavirus enters a human cell and unravels its genome. This transforms the virus from a hardy infectious particle, which must move through the air and evade immune cells, into a naked and vulnerable template, unspooling itself for evaluation. The coronavirus “is a shape-shifter,” Hadden-Perilla said. But scientists don’t fully understand how the virus can tell when it’s time to disrobe.
Perilla said he suspected that some sort of signal inside human cells might trigger the virus’ shell to pop open and release its RNA. That’s the case for the Ebola virus, he said: “It wants to open.” Stopping that process could be vital to halting an infection before it spirals out of control.
A more viral vaccine
In his laboratory in Los Angeles, Gelbart is tackling a construction project of his own: the next great coronavirus vaccine.
His vaccine, designed in collaboration with virus expert Otto Yang, contains two main ingredients, both inspired by ultrasimple viruses that consist of only RNA and protein. The first is an empty protein shell, derived from a harmless plant virus, studded with ready-made coronavirus spike proteins. This viruslike particle is, in a sense, a caricature of the coronavirus. It is not infectious in itself, but can teach the immune system to recognize the actual virus and fight it off, should it try to invade the body.
The Gelbart team’s vaccine also includes a second, spike-free virus shell containing RNA that can instruct human cells to churn out a second wave of coronavirus proteins.
Shots developed by the companies Pfizer-BioNTech and Moderna use similar technology, and are already finding their way into arms around the world. But the RNA in these vaccines is fragile, capable of persisting for only a couple of days after injection, which limits the period when the immune system is exposed to the coronavirus’ spike protein.
To prolong the RNA’s tenure in the body, Gelbart’s next-generation vaccine comes packaged with another molecule to copy the genetic material a few times over, with the hope that the body will both strengthen and lengthen its memory of the coronavirus. Early experiments in the lab suggest that the team’s vaccine, which is still in its infancy, appears to elicit a more vigorous response from certain immune cells. No infectious virus would be produced, only a lengthy lesson for immune cells to learn from.
At the heart of his team’s idea is the ingenuity of viruses, which have already found success at entering their hosts unharmed and delivering their contents to cells. That process, Gelbart said, doesn’t need much modification to become a safe and sustainable option to ward off future disease.
“We’re learning from the virus,” he said. “How to protect RNA, and get it where you want it.”
For him and others in the field, the simplicity of viruses is the source of their appeal, and their strength — one that humankind has yet to fully match.
“Cells want to kill them, societies want to kill them,” Perilla, of the University of Delaware, said. “And yet, they survive.”
By Katherine J. Wu
c.2021 The New York Times Company
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!
Find the best of Al News in one place, specially curated for you every weekend.
Stay on top of the latest tech trends and biggest startup news.