


Highlights: - Indian pharma impacted by regulatory headwinds for Ranitidine - Formulators/API manufacturers to test data within 30 days - Indian formulators can have sales impact of ~5 percent if ban imposed - API manufacturers such as SMS Lifesciences, Solara Active, Dr Reddy’s Lab, Saraca Lab impacted - Ranitidine contributes 5% of Solara’s revenue; company has a presence in Ranitidine’s alternative - Minor impact on Amine manufactures – Ranitidine intermediate DMA HCL has multipurpose usages -------------------------------------------------
Some Indian pharma and API companies have been impacted by the news flow regarding a potential ban on popular drug used for acid reflux, ranitidine.
The US FDA has learned that few of ranitidine products contain a nitrosamine impurity called N‐nitrosodimethylamine (NDMA), a potential carcinogenic.
This has led to a halt in production across the globe starting with Novartis, which issued a product recall. The US FDA has asked drug formulators and API manufacturers to provide test data on ranitidine ingredients/related APIs in a stipulated period of about 30 days.
While the impact on formulators is direct, API and chemical manufacturers can partially offset if they have a presence in alternative drug and applications.
Impact on pharma companies
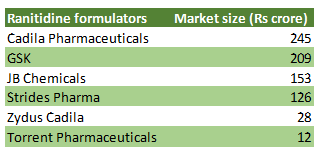
The global formulation market of ranitidine is estimated to be about Rs 20,000 crore. The impact of the development on Indian drug formulators varies, but at an aggregate level, the overall market, which is at stake is about Rs 750 crore.
In the case of GSK Pharma, ranitidine sales opportunity is reported to be ~Rs 200 crore, which is about 6.5 percent of the company's FY19 sales. For Strides Pharma, global ranitidine sales were ~US$ 9 million in H1FY20, or about 4 percent of its FY19 sales.
Select API companies have been identified for more actions than just asking for test data at this point of time. For instance, Saraca Laboratories Limited, which supplies APIs to GSK Pharma, has been notified by the European Directorate for V Quality of Medicines that its certificate of suitability for ranitidine hydrochloride API has been suspended.
GSK Pharma has issued a product recall for medicines having APIs sourced from Saraca Laboratories. Among other API companies impacted are SMS Lifesciences, Solara Active, Dr Reddy’s Lab etc. Note that SMS Life Science is one of the top three suppliers of ranitidine hydrochloride API globally.
Table: Key ranitidine formulators in India
Takeaway for investors
Among the companies under our coverage, there is an impact on Solara Active Pharma Sciences. The company has temporarily suspended production and distribution of ranitidine hydrochloride drug substance.
The ingredient is one of the top 10 APIs for the company contributing about 5% to the overall revenue. Solara Active mostly supplies to its parent company - Strides Pharma - as far as this API is concerned.
The saving grace for the company is that it has presence in one of the alternatives for ranitidine hydrochloride. It manufactures pantoprazole sodium, which belongs to a class of drugs known as proton pump inhibitors.
Still, we believe there would be some impact on earnings for FY20, wherein the range would depend upon whether it is a temporary sales disruption or a product recall. Investors should wait for the test result and regulatory action, if any, before acting.
Among stocks that were severely impacted on Friday was Strides Pharma promoter group company Sequent Scientific. We believe the market reaction was unwarranted given that this company has no direct impact of ranitidine controversy.
Sequent is the largest animal healthcare company in India and has had reported strong operating result recently. However, a word of caution for the investors is that 38 percent of the promoter’s shareholding value is pledged. Thus, the company can have a negative rub-off from the events in Strides Pharma.
Among stocks that can have a minor impact are amine manufacturers. Both Alkyl Amines and Balaji Amines are key manufacturers of DMA HCL (Dimethylamine hydrochloride), an ingredient mostly used as an intermediate for ranitidine. Having said that impact is expected to be limited as DMA HCL has multiple applications, including drugs for diabetics (Metformin), opioid pain medication (Tramadol), high blood pressure and coronary artery disease (amlodipine).
Pls read: Balaji Amines: Growth to pick up in H2
We remain positive on the business fundamentals of amine manufacturers and Sequent Scientific. For Solara Active, we are closely tracking the developments.
The ranitidine episode once again highlights that with the increasing opportunity of Indian manufacturers in the entire global supply chain of fine chemicals/API/pharma, there also lies the growing need to maintain safety and regulatory compliances, which can help them differentiate.
Another similar product recall which global pharma is grappling with for the last one year is that of blood pressure drug valsartan. In India, pharma companies such as Aurobindo, Torrent, Hetero labs were impacted. To draw a comparison, the valsartan market in India is pegged at Rs 260 crore.
Another key takeaway from this is that companies, which are reasonably diversified in terms of exposure to formulations/APIs have a better chance to survive regulatory crises like this.
For instance, SMS Life science almost drives 50 percent of revenue from the Ranitidine HCL API. This explains the over 40 percent correction in the stock price following this episode. Solara Active is relatively better placed with a portfolio of about 50 APIs.
For more research articles, visit our Moneycontrol Research page
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!
Find the best of Al News in one place, specially curated for you every weekend.
Stay on top of the latest tech trends and biggest startup news.