



Numerous studies are carried out at CILICANT to understand the processes behind the challenges faced by formulators and manufacturers when it comes to pharmaceutical packaging. It’s only by doing so that we can develop products that tackle issues that cause the degradation of pharmaceutical products successfully.
One area in which we have invested considerably is in studies related to water activity, or equilibrium relative humidity (ERH), as it is more commonly known. As a result, we have discovered that the use of relative humidity regulators is the best desiccant choice where water activity must be maintained within a narrow range.
To understand why, we need to look at the science behind it.
Defining and determining water activity
Water activity is defined as the ratio of the vapour pressure of water in a given sample (P) to the vapour pressure of pure water (P0) at the same temperature. The formula used is:
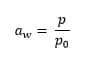
Pure water has a water activity of 1.0, while other substances fall along a sliding scale from this upper limit towards a water activity value of 0.0, indicating complete dryness. When it comes to packaged pharmaceuticals, it’s more convenient to express water activity in terms of the equilibrium relative humidity (ERH) of the sealed system. The equilibrium relative humidity expresses the water activity as a function of the packaging environment and the relationship between the two is represented by the following equation:

Next it’s important to understand the difference between ‘water activity’ and ‘water content’, as they are different concepts. Water (or moisture) content of a pharmaceutical is typically used to refer to the percentage of water molecules in a product, while water activity is a measure of how reactive those water molecules are.
Although there is no direct formulaic approach to plotting the relationship between water content and water activity, the changes in the relationship between water content and water activity is known as a ‘moisture sorption isotherm’ and can be determined for each pharmaceutical product. As the composition of the product changes so too will the moisture sorption isotherm.
There are several ways to determine the water activity of pharmaceuticals. The dew point or chilled mirror method is the gold standard. Here, specialised instruments measure the temperature at which air (at moisture equilibrium) condenses on a polished, chilled mirror exposed to a test sample of the product. This temperature is referred to as the dew point and determines the estimated relative humidity of the sample. Other approaches involve the use of capacitance hygrometer sensors or resistance hygrometer sensors.
Water activity and protecting pharmaceuticals
In the terms of the pharmaceutical industry, water activity is an important metric as it tells us how much moisture is available in a pharmaceutical product for reacting with other substances. Unlike moisture content, water activity accounts for the energy levels of this water, which will impact on the shelf-life of pharmaceutical products. Since pharmaceuticals have different water activity values, by measuring water activity we can evaluate how resistant a pharmaceutical product will be to microbial contaminants. After that, we can assess the best ways to package products in order to maintain a water activity range that is optimal against microbial growth.
Of course, pharmaceuticals come in a combination of formats, such as gel capsules and powder, depending on the most effective delivery mechanism to induce the effects of the medication on the human body. The key is the difference in water activity between the two materials. As moisture moves from a region of high water activity towards one of low water activity, the disparity can lead to issues, such as the cracking of gel capsules or an increase of water activity in the powder.
Many pharmaceutical packaging solutions incorporate the use of one or more desiccants to regulate moisture levels. Together, these aim to bring about low water activity to protect products against the microbial proliferation and other degradation issues due to moisture build-up. However, where the ERH of the packaging environment must be maintained within a specific range, these ‘standard’ desiccants may cause over-desiccation.
Introducing ACCUFLIP: a better way to regulate water activity
CILICANT has addressed this issue by creating a new range of products – ACCUFLIP. These moisture-regulating sorbents, or humidity regulators, are a new type of desiccant designed to maintain a stable ERH within the packaging. Unlike traditional standard desiccants that can be too aggressive when reducing moisture, moisture-regulating sorbents maintain the ERH of the packaging at an optimum level, allowing for an optimal shelf-life. Furthermore, these moisture-regulating sorbents can be adjusted to meet the requirements of specified ERH levels, making them highly versatile and ideal for a wide range of pharmaceutical packaging scenarios.
ACCUFLIP applications
Since humidity regulators can regulate water activity in all forms of pharmaceuticals, the application potential is huge.
For instance, in gel capsules, the water activity of the exterior coating needs to be maintained at a level that prevents the coating becoming brittle as a result of very low ERH, or becoming sticky as a result of high ERH. As moisture-regulating sorbents take up and release moisture as required to maintain a constant ERH, the gel capsules are protected from both high and low ERHs. A standard desiccant would be ineffective in this instance as it would aggressively remove moisture from the environment, potentially leading to a very low ERH and low water activity, resulting in capsule brittleness.
Another application can be seen with dry powder inhalers, widely used for delivering of drug doses to the lungs. It’s essential that these devices are packed in an environment with a stable ERH as moisture fluctuations have negative effects on the stability of the product, detrimentally impacting efficacy for end-users. A high ERH here would result in powder particles clumping together making efficient drug delivery to the lungs problematic. Here, moisture-regulating sorbents prevent moisture levels becoming too high or too low, where standard desiccants would over-desiccate and create electrostatic charges, potentially lowering the dose available to lung tissue.
ACCUFLIP – a new tool in pharmaceutical packaging
Clearly, the importance of a well-regulated water activity in the stability of many pharmaceutical dosage forms cannot be over-emphasised. While traditional, standard desiccants can lower water activity in packaging environments and extend the shelf-life of pharmaceuticals, they can also lead to over-desiccation. In these scenarios, moisture-regulating sorbents, such as ACCUFLIP, provide an effective alternative that helps to keep the ERH of packaged pharmaceuticals at an optimal level for longer shelf stability.
At CILICANT, we’re always looking at new and innovative ways of solving problems for our clients in the pharmaceutical industries and believe that these desiccants will play a vital role in the pharmaceutical packaging industry as more products requiring strict ERH levels come to market.
To see the full white paper related to this article, visit www.cilicant.com
The information provided by Cilicant Chem Private Limited in this paper is for general informational purposes only and shall not be relied upon by anyone for any other purpose. We make no representation or warranty of any kind, express or implied, regarding the accuracy, adequacy, validity, reliability or completeness of any information provided in this paper. Cilicant Chem Private Limited owns all the intellectual property rights in the contents of this paper. The contents of this paper shall not be replicated or reproduced in any manner whatsoever without the prior written permission of Cilicant Chem Private Limited. Cilicant Chem Private Limited shall not be liable for any direct, indirect, incidental, consequential or punitive damages or losses resulting from your reliance on the data provided in this paper."
Moneycontrol journalists were not involved in the creation of the article
Discover the latest Business News, Sensex, and Nifty updates. Obtain Personal Finance insights, tax queries, and expert opinions on Moneycontrol or download the Moneycontrol App to stay updated!