This LIVE blog session has concluded. Stay tuned to Moneycontrol for the latest news, views and updates.
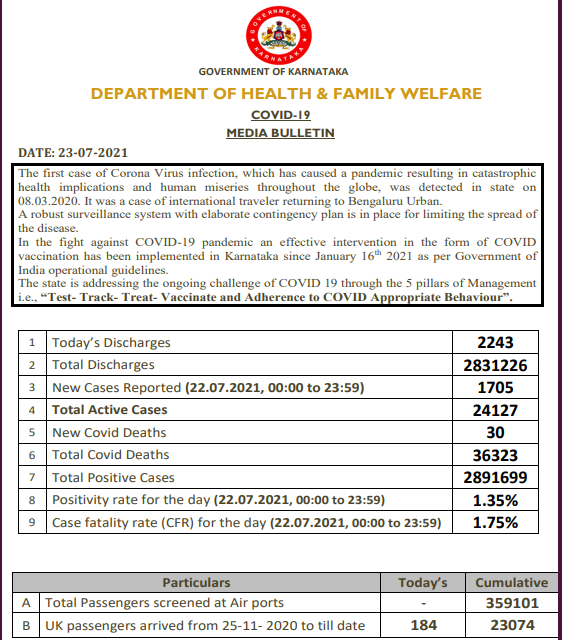
Coronavirus News Highlights: With a single-day rise of 35,342 cases, India's COVID-19 tally climbed to 3,12 93,062 on Friday, while 483 more fatalities pushed the death toll due to the viral disease to 4,19,470, according to the health ministry. The number of active cases has declined to 4,05,513, accounting for 1.3 percent of the total caseload, while the national COVID-19 recovery rate was recorded at 97.36 percent, the ministry's data updated at 8 am showed. A decline of 3,881 cases was recorded in the active COVID-19 caseload in a span of 24 hours. Also, 16,68,561 tests to detect the infection were carried out on Thursday, taking the total number of such tests conducted so far in the country to 45,29,39,545, while the daily positivity rate was recorded at 2.12 percent. It has been less than three percent for 32 consecutive days, the ministry said. The weekly positivity rate was recorded at 2.14 percent. The cumulative number of Covid vaccine doses administered so far in the country has reached 42.34 crore. India's COVID-19 tally had crossed the 20-lakh mark on August 7 last year, 30-lakh mark on August 23, 40-lakh mark on September 5 and 50-lakh mark on September 16. It went past 60 lakh on September 28 last year, 70 lakh on October 11, 80 lakh on October 29, 90 lakh on November 20 and the one-crore mark on December 19. India crossed the grim milestone of two crore Covid cases on May 4 this year and the three-crore mark on June 23.
This LIVE blog session has concluded. Stay tuned to Moneycontrol for the latest news, views and updates.
Scientists of reputed research institutions of the country have found AYUSH-64, a polyherbal formulation, helpful in the treatment of asymptomatic, mild and moderate COVID-19 infection as an adjunct to standard care, the government said on Friday.
In a written response to a question in Lok Sabha, Minister of State for AYUSH Dr Mahendra Munjapara said the Ministry of Ayush-Council of Scientific and Industrial Research (CSIR) collaboration has recently completed a robust multi-centre clinical trial to evaluate the safety and efficacy of AYUSH-64 in the management of mild to moderate COVID-19 patients.
Bharat Biotech announces termination of its MOU with Precisa Medicamentos & Envixia Pharmaceuticals to sell COVAXIN in Brazil. "We'll continue to work diligently with ANVISA, the Brazilian drug regulator to complete the regulatory approval process for the vaccine," said thebiotechnology company.
India’s cumulative COVID vaccination has exceeded 42.75 crore till 7pm today. Nearly 39 lakh doses have been administered on July 23, says governmnet.
Madhya Pradesh on Friday recorded 11 COVID-19 cases that raised its tally of infections to 7,91,732, an official from the state health department said.
The count of recoveries reached 7,81,048, while the toll stood at 10,512, as no fresh fatalities were reported, the official said, adding that the central state currently has 172 active cases.
With the addition of 73,709 swab samples tested during the day, the total number of tests conducted in the state rose to 1,38,53,243, the official said.
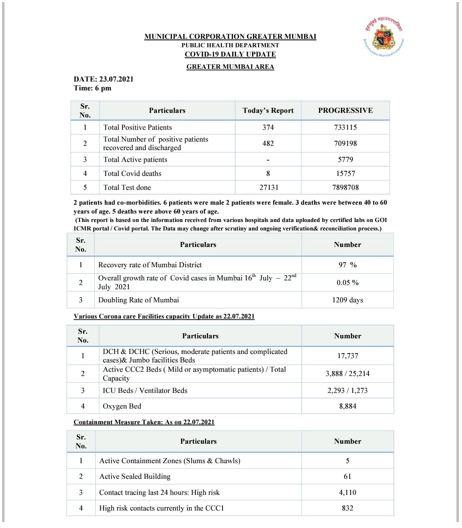
Goa's coronavirus caseload went up by 73 and reached 1,70,272 on Friday, while 139 patients recovered from the infection, a health department official said. The death toll rose to 3,126 as three more patients succumbed to the infection during the day, he said. The number of recovered cases increased to 1,65,839 after 139 patients were discharged from hospitals in the last 24 hours, the official said. Goa now has 1,307 active cases. "With 3,031 new tests, the total number of tests conducted in the state has gone up to 10,21,344,” he added. Goa's COVID-19 figures are as follows: Positive cases 1,70,272, new cases 73, death toll 3,126, discharged 1,65,839, active cases 1,307, samples tested till date 10,21,344.
The European Medicines Agency has recommended approving Moderna's COVID-19 vaccine for children aged 12 to 17, the first time the shot has been authorised for people under 18.In a decision on Friday, the EU drug regulator said research in more than 3,700 children aged 12 to 17 showed that the Moderna vaccine — already given the OK for adults across Europe — produced a comparable antibody response.Until now, the vaccine made by Pfizer and its German partner BioNTech has been the only option for children in North America and Europe.The US Food and Drug Administration is currently considering whether to extend the use of the Moderna vaccine for children under 18.With global vaccine supplies still tight, much of the world still is struggling to immunize adults. US and European regulators do caution that both the Moderna and Pfizer vaccines appear linked to an extremely rare reaction in teens and young adults — chest pain and heart inflammation.
About 100 of the 613 U.S. athletes descending on Tokyo for the Olympics are unvaccinated, the U.S. Olympic and Paralympic Committee's medical chief said hours before Friday night's opening ceremony.Medical director Jonathan Finnoff said 567 of the American athletes had filled out their health histories as they prepared for the trip, and estimated 83% had replied they were vaccinated.Eighty-three percent is actually a substantial number and we're quite happy with it, Finnoff said.Nationally, 56.3% of Americans have received at least one dose of the vaccine, according to the Centers for Disease Control and Prevention.
Jharkhand reported 22 new COVID-19 cases in the last 24 hours, which pushed the tally to 3,46,846, a health department bulletin said on Friday.The COVID-19 death toll remained at 5,122 as no new fatality due to the contagion was reported in the last 24 hours, it said.Of the 22 new cases, a maximum of 5 new cases were reported from Deoghar followed by three cases from state capital Ranchi and two cases each from Dhanbad, Koderma, Ramgarh, Saraikela and Simdega.No new COVID-19 case was reported from Chatra, Dumka, Garhwa, Giridih, Godda, Gumla, Hazaribag, Jamtara, Khunti, Latehar, Lohardaga, Pakur, Sahebganj and West Singhbhum.Jharkhand now has 305 active COVID-19 cases, while 3,41,419 patients have recovered from the disease including 32 during the last 24 hours, the bulletin said.The mortality rate in the state remained at 1.47 per cent, higher than the national average of 1.30 percent.
The demand for medical oxygen in the country peaked to nearly 9,000 MT in the second Covid wave as compared to the peak requirement of 3,095 MT during the first wave, Lok Sabha was informed on Friday.To a question on shortage of medical oxygen in the country during the second wave of the coronavirus pandemic, Union Minister Bharati Pravin Pawar said supply of medical oxygen to hospitals is determined by contractual arrangements between the hospital concerned and the supplier."However, due to an unprecedented surge in demand of medical oxygen across the country during the second wave of COVID-19 -- the demand in the country peaked to nearly 9,000 MT (metric tonnes) as compared to the peak requirement of 3,095 MT during the first wave," she said in a written reply.It was in this context that the central government stepped in and took all possible measures to increase production and to facilitate equitable distribution to states, the minister of state for health said.
Some states had floated global tenders for COVID-19 vaccines in May but none of these materialised into procurement, Union Health Minister Mansukh Mandaviya informed Lok Sabha on Friday.Multiple and repeated demands were made by states and union territories to the government of India that they be allowed to procure vaccines directly and administer them as per their requirements, and such global tenders were placed by them in May 2021, Mandaviya said in a written reply."However, none of the tenders floated by any state government materialised into procurement of Covid vaccines from any vaccine manufacturers, including offshore manufacturers," he said.Later on, many states communicated to the government of India that they were facing difficulties in managing the funding and logistics of direct procurement of Covid vaccines."This in turn impacted the pace of the National COVID-19 Vaccination Program. Keeping in view the repeated requests received from states, and the experience gained, the Guidelines for National COVID Vaccination Program were reviewed and ''Revised Guidelines for Implementation of National COVID Vaccination Program'' were issued on June 8, 2021, to be effective from June 21," he said in the written reply.